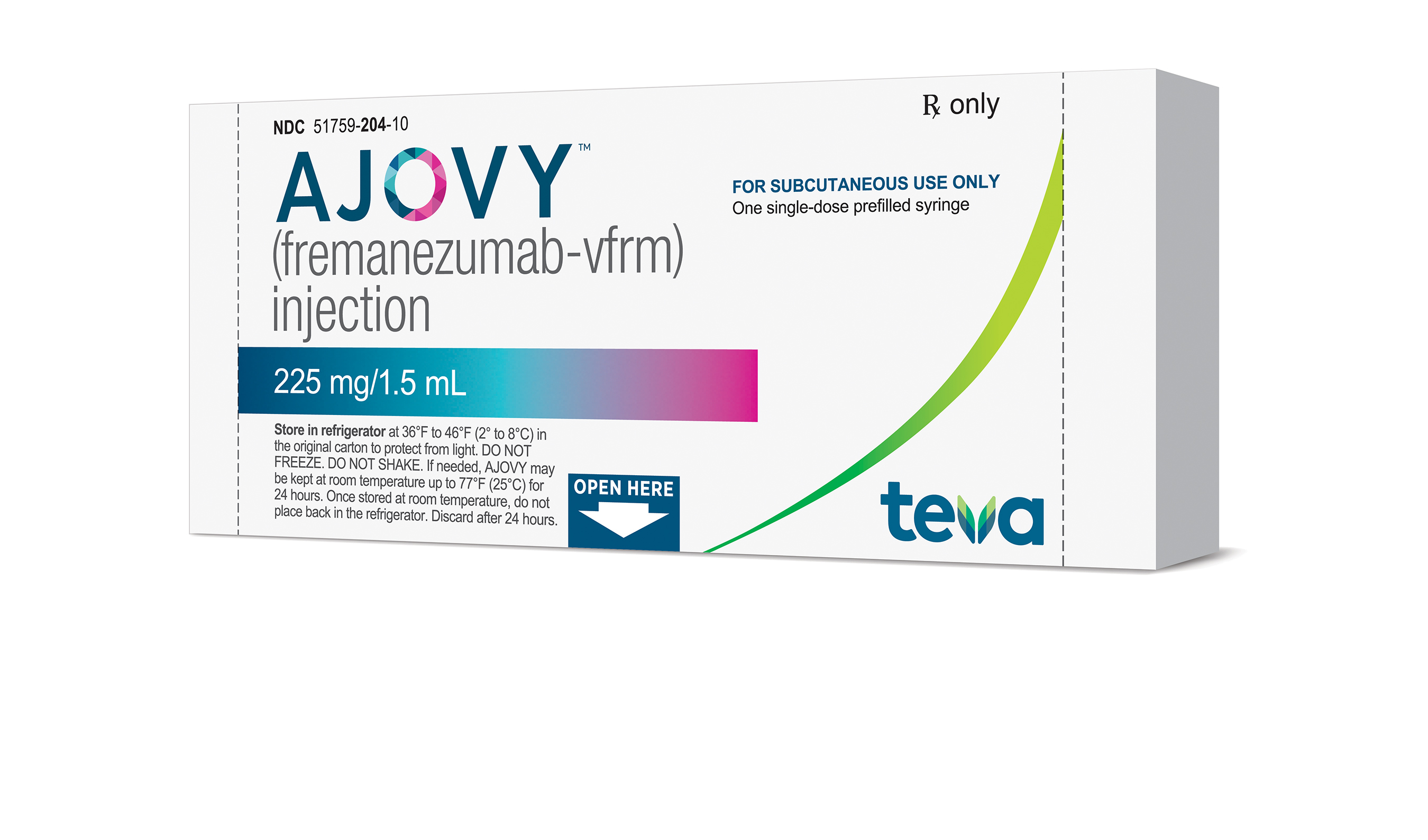
ADDING MULTIMEDIA Teva Announces U.S. Approval of AJOVYTM (fremanezumab-vfrm) Injection, the First and Only Anti-CGRP Treatment with Both Quarterly and Monthly Dosing for the Preventive Treatment of Migraine in Adults | Business

Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA












/cloudfront-us-east-2.images.arcpublishing.com/reuters/NF44W6QYRFLRZHINMZOWHMD37I.jpg)
/cloudfront-us-east-2.images.arcpublishing.com/reuters/CCBS56SBRRKUDM7JEVVE3UOQEE.jpg)






